Many of us know, or have known, someone with prostate cancer. Today, prostate cancer is seen as a chronic disease and it is common that surgery is not performed in cancers categorized as harmless, mainly due to the high risk of post-surgery complications. I am not alone in saying that current diagnostic methods for deciding whether the cancer should be removed have many flaws—mainly due to a lack of supporting diagnostic IT tools that can assist the radiologist, pathologist and urologist.
Prostate cancer at a glance
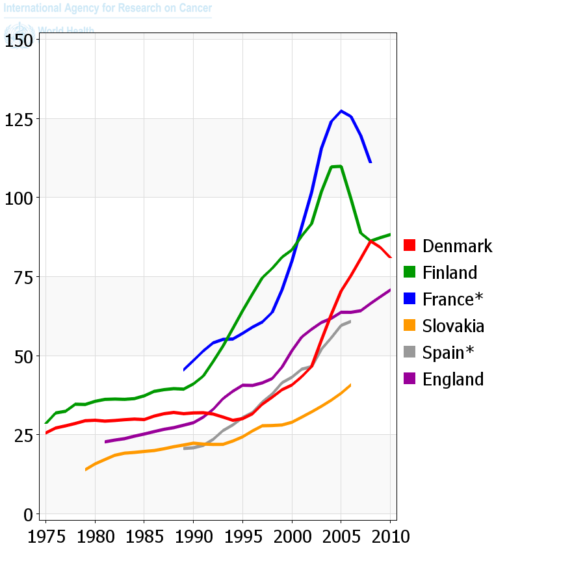
Figure 1. Trends in incidence of prostate cancer in selected countries: age-standardized rate per 100,000 (8).
Prostate cancer is the second most common cancer in men globally. In more developed regions, prostate cancer accounts for almost 70% of cases. In total, prostate cancer is the fifth leading cause of cancer death in men (1).
Because of its high mortality rate, it has become common practice in developed countries to perform prostate-specific antigen (PSA) testing and a subsequent biopsy. The higher availability of PSA screening has led to a surge in prostate cancer incidents (see figure 1), identifying men without any symptoms of the disease. (2).
One issue with PSA testing, however, is that it detects many prostate cancers that are small and/or would otherwise remain unrecognized, which may or may not develop further into a higher stage disease (2). One study concluded that between 11.3 and 19.8% of men with prostate cancer diagnosed through PSA tests will receive a false-positive diagnosis. About half of those will be affected by overdiagnosis, leading to invasive treatment. (3). Due to the lack of accuracy in existing diagnostic methods, many men decide to undergo surgery, which in many cases results in severe complications that lower the quality of life. (2).
According to many professionals, prostate cancer diagnostics are far behind other common cancer types, such as breast cancer, where several countries are conducting successful country-wide screening programs. Similar initiatives are needed for prostate cancer, but current diagnostic methods are unfortunately considered too inaccurate to be beneficial.
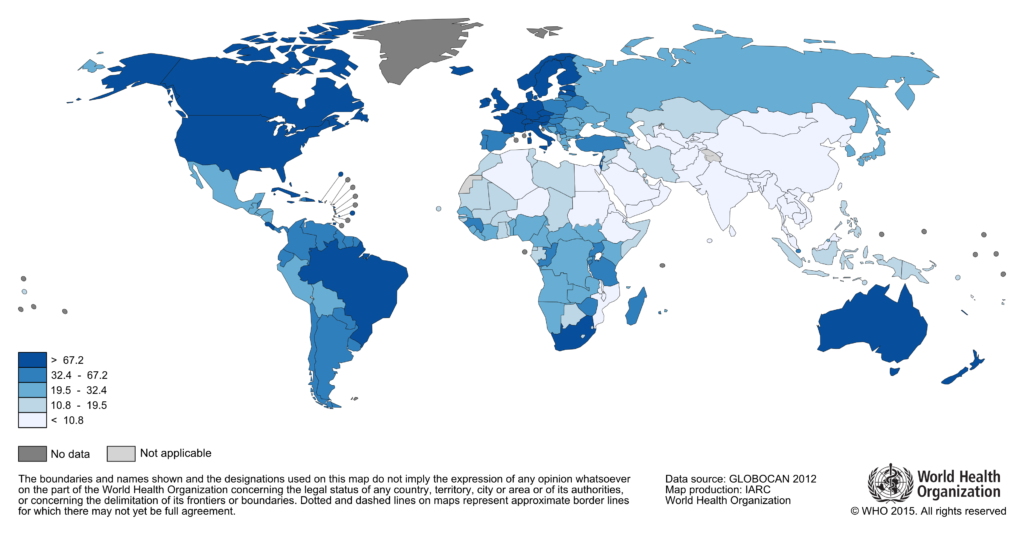
Figure 2. Estimated prostate cancer incidence worldwide in 2012 (8).
Complications with surgery
About half of all people who have prostate cancer surgery experience complications afterwards. For example, 40% suffer from erectile dysfunction due to nerve damage during surgery, and two out of 100 men experience major problems with incontinence one year after their surgery. (2).
The high frequency of complications is concerning since most prostate cancers are slow growing and harmless. The risk assessment of how life threatening the cancer is therefore represents an important aspect of the treatment decision and should be weighed against the risk of surgery complications. (3).
Surgery complications have led to a passive strategy called “active surveillance” or “watchful waiting.” Doctors carefully monitor the prostate cancer for signs of growth and progression using blood tests and other exams. This helps avoid the urinary and erectile complications associated with certain treatments. (3).
Overall, there is an urgent need for better ways to diagnose and risk-assess prostate cancer to identify and sort out the dangerous cases from the harmless ones.
Challenges in diagnosis
To further judge whether the cancer is dangerous and should be removed—or only be observed—the standard practice is to perform several biopsies from the prostate according to a standardized protocol. These biopsies, typically comprising 10–12 core template biopsies, are judged and graded by a pathologist, often using the Gleason pattern (see figures 3 and 4).
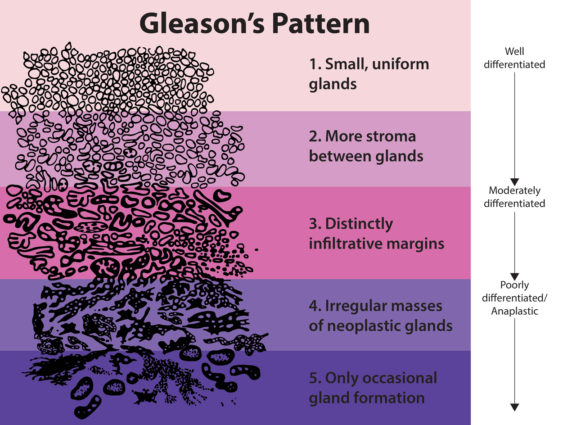
Figure 3. The Gleason scoring pattern (7).
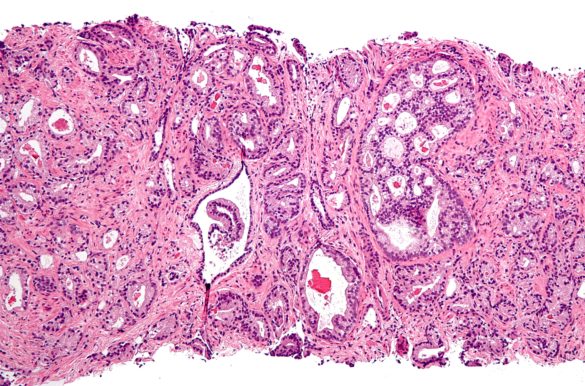
Figure 4. Needle biopsy from prostate stained with H&E (7).
The biopsy procedure has several shortcomings. One is a high rate of inaccurate biopsies, resulting in an overdetection of insignificant prostate cancers and an underdetection of significant disease. The scoring according to the Gleason pattern is also time consuming if it is not done using digital pathology and image analysis, and its correlation to severity of disease and clinical relevance is heavily debated. (4).
The current biopsy procedure involves a urologist taking transrectal ultrasound-guided biopsies. This means that in modern care systems, 3D imaging is used to visualize the area where the biopsies have been taken to ensure the suspected lesion is sampled. Many still claim it is difficult to ensure that the tumor is hit and that the biopsies are representative.
Once the biopsies are taken by the urologist and judged by the pathologist, the multidisciplinary team meeting (MDT) or tumor board is an important forum for deciding the subsequent treatment of the patient. This meeting involves the radiologist, pathologist, surgeon, urologist, and other healthcare professionals.
In the MDT, one major challenge is to use the MRI images to judge where each biopsy was taken in order to map out the location of the tumor. Cases can become extremely complex when several tumors are present. The correlation between the biopsies and the area where they were taken is highly important information for guiding the surgery and avoiding complications by not damaging the surrounding nerves. Unfortunately, most PACS (Picture Archiving and Communication System) used in the MDT lack tools to facilitate biopsy and MRI matching.
The advancement of MRI in prostate cancer diagnostics
This shortcomings in the biopsy procedure have led to a debate as to whether an MRI should be performed prior to the biopsy to better judge if a biopsy is necessary or not—as a kind of risk assessment.
There is compelling research-based evidence showing that mpMRI (multi-parametric magnetic resonance imaging), which combines three types of scans to create a clearer picture, can help determine if a biopsy is necessary or not. (5), (4).
Another advantage of an MRI is that if a suspicious area is detected, a real-time MRI-guided targeted biopsy facilitates pinpoint diagnosis and treatment matching.
These benefits have been demonstrated in a recently published study of 500 men with raised PSA levels who had not undergone biopsy. The study compared the standard of care using transrectal ultrasound-guided biopsy with MRI. The result: The use of risk assessment with MRI before biopsy and MRI-targeted biopsy was superior to using ultrasound (see figure 5). (5).
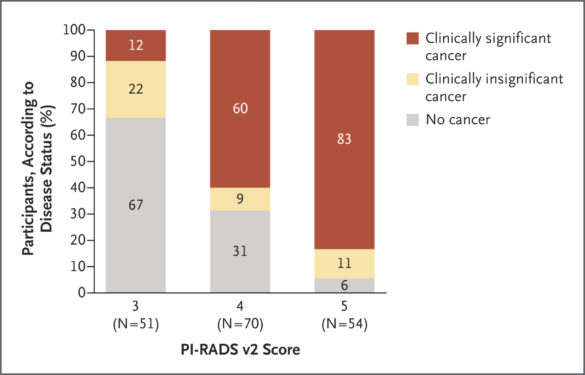
Figure 5. The percentage of men with clinically significant, clinically insignificant, and no cancer, identified according to PI-RADS v2 score. Showing that MRI led to higher accuracy (6).
I truly believe that we will see an increased use of MRI in prostate cancer diagnostics, both before the biopsy in the risk assessment, assisting the biopsy procedure itself, and post biopsy for accurate positioning mapping. This, in turn, will lead to a need for change in the care process in terms of how those images and findings are discussed among all physicians in the care team during the MDT.
The road ahead
MRI is advancing in prostate cancer diagnostics and there is an obvious need, not only for pre-operative tools to estimate risk, but also follow-up and correlation tools to help determine where in the images each biopsy is taken. The most obvious system to host these tools is the PACS, and I am sure that we will soon see vendors making these available on the market.
Hospitals handling digital pathology images in the same PACS are likely to be the first to take advantage of prostate cancer diagnostic tools. This is due to the tight integration between images from the biopsy, the MRI and the ultrasound. If the PACS also supports structured reporting for both radiology and pathology, it could also include tools for standardized interpretation and reporting, such as PIRADS. Due to the need for improvement in prostate cancer diagnostics, and the advancements in using MRI for diagnosis, I am confident that we will see a great deal of improvements in the near future. It would be odd if this revolution in prostate cancer diagnostics did not take place in the PACS.
References
- World Cancer Research Fund International. Prostate cancer statistics. [Online] 01 16, 2015. [Cited: 03 13, 2018.] https://www.wcrf.org/int/cancer-facts-figures/data-specific-cancers/prostate-cancer-statistics.
- Cancer Research UK. Problems after surgery. [Online] 07 06, 2016. [Cited: 03 13, 2018.] http://www.cancerresearchuk.org/about-cancer/prostate-cancer/treatment/surgery/after-surgery/problems.
- Medical Daily. PSA Test Deemed Too Inaccurate, Unspecific For Prostate Cancer Screening Guidelines. [Online] 10 27, 2014. [Cited: 04 12, 2018.] https://www.medicaldaily.com/psa-test-deemed-too-inaccurate-unspecific-prostate-cancer-screening-guidelines-308018.
- Prostate.net. Should I Do A Multiparametric MRI Before Having A Prostate Biopsy? [Online] 05 18, 2017. [Cited: 03 22, 2018.] https://prostate.net/articles/should-i-do-a-multiparametric-mri-before-having-a-prostate-biopsy.
- Healthline. Prostate Cancer Complications. [Online] [Cited: 03 20, 2018.] https://www.healthline.com/health/prostate-cancer-complications.
- MRI-Targeted or Standard Biopsy for Prostate-Cancer Diagnosis. Veeru Kasivisvanathan, M.R.C.S., Antti S. Rannikko, Ph.D., Marcelo Borghi, M.D., Valeria Panebianco, M.D., Lance A. Mynderse, M.D., Markku H. Vaarala, Ph.D., Alberto Briganti, Ph.D., Lars Budäus, M.D., Giles Hellawell, F.R.C.S.(Urol.), Richard G. Hindley,. s.l. : New England Journal of Medicine, 2018, Vol. DOI: 10.1056/NEJMoa1801993. http://www.nejm.org/doi/full/10.1056/NEJMoa1801993.
- Wikipedia on Gleason Scoring Pattern. Wikipedia. Gleason Scoring Pattern. [Online] [Cited: 03 29, 2018.] https://en.wikipedia.org/wiki/Gleason_grading_system.
- World Health Organization. International AGency for Research on Cancer. Prostate cancer . [Online] 3 11, 2018. http://globocan.iarc.fr/old/FactSheets/cancers/prostate-new.asp.
